
The medical field is the primary application for the argon laser system, as it can target areas with exceptional precision. Argon lasers are employed in the treatment of diabetic retinal detachment and diabetic phototherapy, as well as in surgeries to fuse arteries and eliminate tumors.The potassium entry describes this method of dating and determining the age of rocks. However, one non-radioactive isotope can be used to determine the age of extremely old rocks. There are no practical uses for radioactive argon isotopes. These particles cling to the atoms, making them radioactive. When tiny particles are fired at atoms, radioactive isotopes are created. Each isotope element represents a change.Ī radioactive isotope breaks apart and emits radiation when it decays. 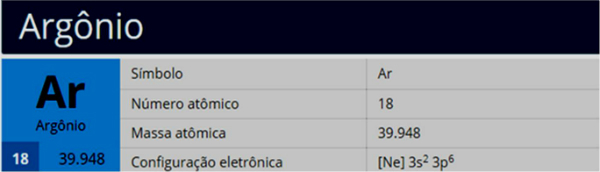
The element is defined by its number of protons, however, the number of neutrons in an atom of any given element can vary. The number of protons plus neutrons in the nucleus of an element's atom is used to calculate its mass number. The mass number is the number printed to the right of the element name. Isotopes differ from one another based on their mass number. Isotopes are formed when an element splits into two or more isotopes. Argon-36, Argon-38, and Argon-40 are the isotopes in question. The three isotopes of argon are found in nature. Some argon can be recovered as a by-product of the natural gas purification process. Argon can also be found in natural gas wells. It does not react with magnesium or calcium, therefore it stays behind. With nitrogen gas, a little amount of argon is always present as an impurity. A nitride is formed when magnesium or calcium reacts with nitrogen: Argon can also be made by combining heated magnesium or calcium with nitrogen gas from the atmosphere. The noble-gas mixture Fractional distillation can be used to create argon from liquid air. It is commercially generated via fractional distillation, which separates it and other gases from liquid air. When naturally occurring 40K (radioactive potassium) decays to 40Ar, it produces 40Ar. 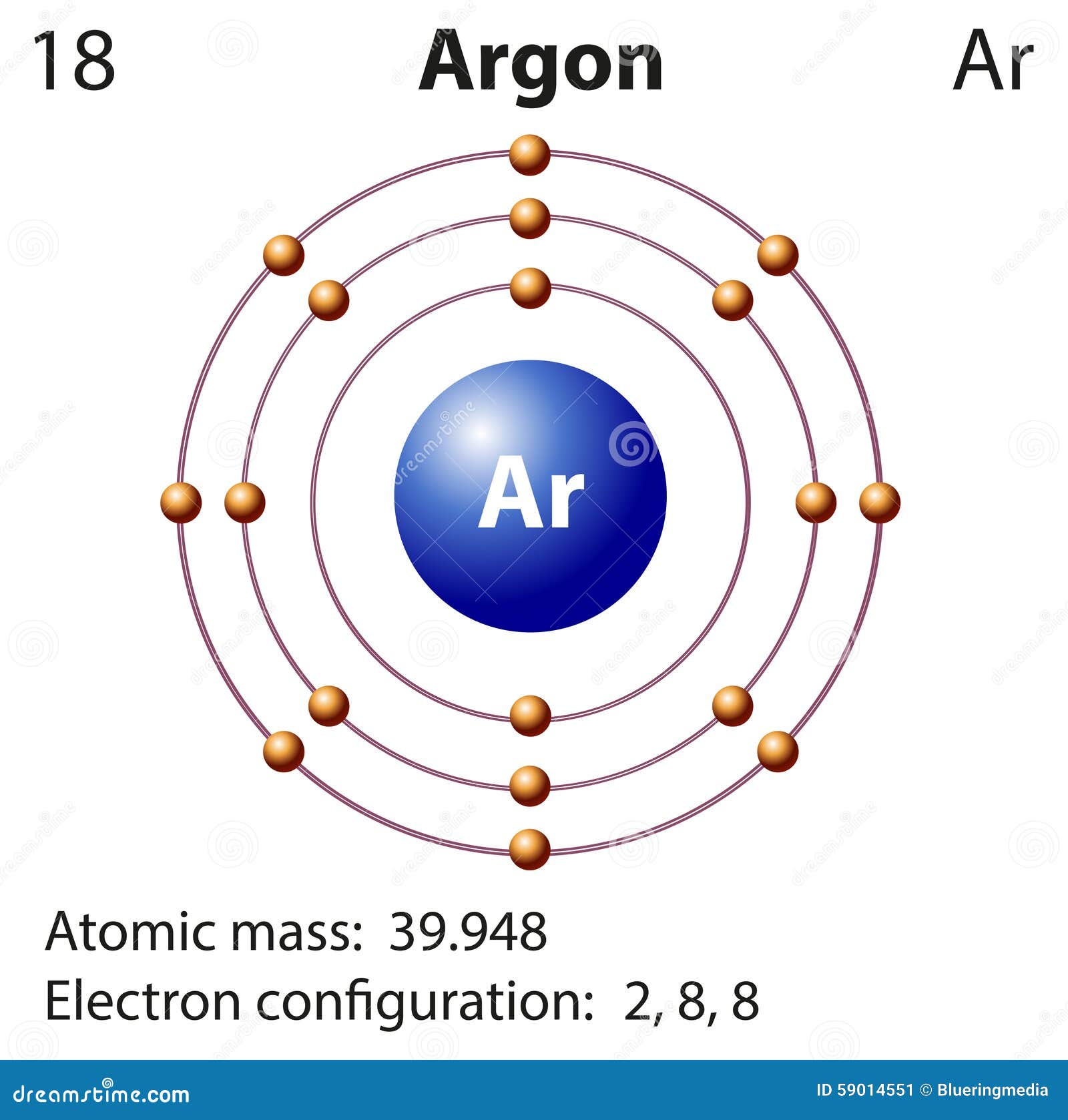
Ramsay and Rayleigh announced their discoveries to the rest of the world in 1895.Īrgon is the third most abundant gas in the atmosphere, accounting for 0.94 percent of the Earth's atmosphere. 
Ramsay discovered new red and green spectral lines after analyzing the spectrum. The gas that remained was chemically inert and occupied 1/80th of the original volume. The nitrogen in the gas was eliminated by reacting it with magnesium, yielding solid magnesium nitride. Ramsay isolated all of the components of the air, including nitrogen, oxygen, and carbon dioxide, in 1894. Rayleigh and Ramsay conducted additional tests to understand this anomaly and kept each other updated on their progress. Lord Rayleigh discovered in 1893 that nitrogen taken from the air has a higher density than nitrogen extracted from ammonia or other nitrogen compounds. Argon was undetected for many years despite being prevalent in the Earth's atmosphere until William Ramsay and Lord Rayleigh isolated it from liquid air in 1894. However, he was unable to identify the gas and completely comprehend its qualities. For a proton or a neutron, A = 1.In 1785, British scientist Henry Cavendish succeeded in isolating the gas and found that it makes about 1% of air, remaining unreactive even in severe temperatures. Like the notation used to indicate isotopes, the upper left superscript in the symbol for a particle gives the mass number, which is the total number of protons and neutrons. \): Nuclear Decay Emissions and Their Symbols Identity
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
